Basic and Applied R&D
- Advanced Live-Cell Array imaging platforms for long-term multi-parametric analysis of food-grade nanoparticle safety
- The cationic dye basic orange 21 (BO21) as a potential fluorescent sensor
- Microenvironment Mechanics and Live Cell Fate
- Fluctuation of Information Entropy Measures in Cell Image
- Hydrogel microstructure live-cell array for multiplexed analyses of cancer stem cells, tumor heterogeneity and differential drug response at single-element resolution
- Donut-shaped chambers for analysis of biochemical processes at the cellular and subcellular levels
- Monitoring gene expression in individual living cells
- Live cell sensors
- Monitoring changes in membrane potential by calculating energy transfer efficiency via fluorescence polarization measurements
- Differential temporal and spatial biophysical aspects of the fluorescence of cellular fluorophores
- The development and utilization of Fluorescence Resonance Energy Transfer (FRET) imaging based on Fluorescence Polarization (FP) measurements
- Quantitative analysis of tumor metastatic potential at the individual cell resolution via diffusion characteristics
- Computer-controlled Optical tweezers
- Monitoring drug effects and pathological pathways in living cells by non-interfering Optical Tweezers
- Identifying intracellular EGFP-Bid Translocation by quantitative fluorescence measurements
- The biophysical mechanism of FP alteration in EGFP expressed in living cells, following stimulation
- Lysosomal involvement in apoptosis induced by oxidative stress
- Cell-mediated cytotoxicity
Advanced Live-Cell Array imaging platforms for long-term multi-parametric analysis of food-grade nanoparticle safety
As engineered nanoparticles (NPs) are increasingly incorporated into food industry processes and consumer products, ingestion of nanomaterials is inevitable. Studying the potential effects of exposure to these NPs is critical to ensure the health and safety of consumers and the environment.
The harmfulness of ingested NPs depends on their ability to damage cells within both the gastrointestinal tract (GT) and the liver, which are the first organs exposed to ingested NPs. Chronic toxicity is most important since relatively low levels of NPs are likely to be consumed over an extended period.
Currently, there is no consensus regarding the gastrointestinal fate and toxicity of different kinds of inorganic food NPs. There is an obvious need for standardized methods to adequately test food NP toxicity under physiological conditions in a reproducible manner.
In order to meet the above need, the current proposal aims to develop and validate an advanced long-term in-vitro culture system which recapitulates the complex cellular diversity and the function of the native GT and liver. This will be achieved by integrating four features; 3D cell structures, multi cell types, bacterial influence and fluidic stimulus, into a unique imaging platform. Numerous 3D spheroids will be co-cultured with other relevant cell types in a 6-well imaging plate featuring a high optical quality glass bottom patterned with hydrogel array with inlet and outlet tubes, to control the flow above the array. The platform will facilitate simultaneous self-generation of numerous hetero-spheroids, each in a single nL volume micro-chamber, and their concomitant treatment and measurement in a high throughput manner. The spheroids will be exposed to commercial titanium dioxide and silicon oxide NPs, two of the main inorganic NPs that are frequently used in the food industry, and monitored during long term experiments. After treatment with first cycle NPs, spheroid populations will be retrieved, dispersed and re-seeded, to create new generations of 3D structures, enabling investigation of effects of lengthy exposure to food NPs. Dynamic functional analysis of GT and liver cellular models is proposed, utilizing tailored multiplex assays, readouts and image analysis tools for NP uptake, and their effects (e.g oxidative stress, cytotoxicity, GI and liver functionality); concomitantly with dynamic liquid assessment of NP concentrations and soluble secreted factors (e.g cytokines, proteins, enzymes) in medium. The multi-parametric datasets, in combination with unique algorithms, will be used to predict cytotoxicity potential and safety grade of food NPs via machine learning analysis, and to create automatic NP safety assessments. The final validated platform is expected to be adopted by regulatory agencies and the food industry for use in standardized screening to accurately evaluate the toxicological properties of existing and novel food NPs – a platform with high economic potential.
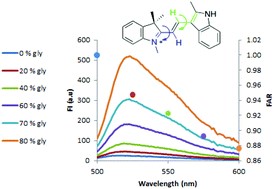
The cationic dye basic orange 21 (BO21) as a potential fluorescent sensor
The present study investigates the fluorescence properties of BO21 and their dependence on various intracellular conditions. The results obtained with cell-free solutions indicate that the influences of pH and temperature on the fluorescence spectra are negligible, while viscosity, various proteins and heparin have significant influence. In the presence of heparin, a red shift of the emission spectrum (from 515 to 550 nm) is observed, suggesting that this shift cannot simply be attributed to electrostatic interaction between BO21 and the polyanionic heparin, but rather to aggregation of BO21 on the polyanion. In water, the quantum yield of BO21 was found to be 1000 times lower than that of fluorescein, yet surprisingly its fluorescence polarization (FP) was found to be about 40 times higher (FP = 0.470), even though both have similar structures and molecular weights. A thorough analytical and experimental investigation of these phenomena indicates that the very high FP of BO21 in water is a consequence of its very short lifetime. However, upon the addition of heparin to aqueous BO21, the fluorescence lifetime (FLT) of BO21 increases from tau = 10.35 to 56.5 ps, with a consequent dramatic drop in its fluorescence polarization from 0.470 to 0.230. From its behavior in aqueous glycerol solution, it is hypothesized, with support from theoretical calculations, that BO21 is a molecular rotor. Using these properties, BO21 may be a good candidate as a sensor, for example, of heparin levels in blood or of intracellular viscosity.
Microenvironment Mechanics and Live Cell Fate
Biological tissues normally possess varying levels of rigidity, which contribute to the performance of their physiological functions. Changes in tissue rigidity may reflect transformation from a normal to a pathological state. Cancer cells and surroundings' feedbacks have mutual effect and regulate the tumor-microenvironment crosstalk causing in the loss of the normal tissue integrity, and support tumor progression and expansion.
This research project benefits by usage of the methodology and device developed in the lab providing an efficient approach to modify the surroundings’ mechanical properties in order to mimic the mechanical interaction in vivo during tissue growth. Different components of live tissue’s that is, cells, water, and scaffold material, can determine whether cells can form cell aggregates and what type of multicellular object is developed. Particularly, cancer cells within the tumor are influenced by the mechanical conditions of their microenvironment, which can drive cell fate. Using physiologically relevant three-dimensional culture system, we studied the 3D cancer microtissues' formation and growth in the conditions of different rigidities.
Non-adherent, non-tethered 3D objects were generated from breast cancer single cells within a hydrogel array, cultured under various mechanical conditions which were created by procedure of agarose embedding, and measured at single-object resolution. Under stiffness that resembles tumor tissue, higher growth rate of breast cancer microtissue and specific “preinvasive” phenotype are evident. Hallmarks of this phenotype include loose morphology with unclear edges and massive peripheral cell spread, dispersed intrinsic structure and reduced expression of epithelial surface markers. Additionally, the control culture of 3D objects grown freely under low-rigidity conditions lacks the specific subset of the pre-invasive phenotype which developed in the stiffer surroundings.
Fluctuation of Information Entropy Measures in Cell Image
A simple, label-free cytometry technique is introduced. It is based on the analysis of the fluctuation of image Gray Level Information Entropy (GLIE) which is shown to reflect intracellular biophysical properties like generalized entropy. In this study, the analytical relations between cellular thermodynamic generalized entropy and diffusivity and GLIE fluctuation measures are explored for the first time. The standard deviation (SD) of GLIE is shown by experiments, simulation and theoretical analysis to be indifferent to microscope system "noise". Then, the ability of GLIE fluctuation measures to reflect basic cellular entropy conditions of early death and malignancy is demonstrated in a cell model of human, healthy-donor lymphocytes, malignant Jurkat cells, as well as dead lymphocytes and Jurkat cells. Utilization of GLIE-based fluctuation measures seems to have the advantage of displaying biophysical characterization of the tested cells, like diffusivity and entropy, in a novel, unique, simple and illustrative way.
Hydrogel microstructure live-cell array for multiplexed analyses of cancer stem cells, tumor heterogeneity and differential drug response at single-element resolution
Specific phenotypic subpopulations of cancer stem cells (CSCs) are responsible for tumor development, production of heterogeneous differentiated tumor mass, metastasis, and resistance to therapies. The development of therapeutic approaches based on targeting rare CSCs has been limited partially due to the lack of appropriate experimental models and measurement approaches. The current study presents new tools and methodologies based on a hydrogel microstructure array (HMA) for identification and multiplex analyses of CSCs. Low-melt agarose integrated with type I collagen, a major component of the extracellular matrix (ECM), was used to form a solid hydrogel array with natural non-adhesive characteristics and high optical quality. The array contained thousands of individual pyramidal shaped, nanoliter-volume micro-chambers (MCs), allowing concomitant generation and measurement of large populations of free-floating CSC spheroids from single cells, each in an individual micro-chamber (MC). The optical live cell platform, based on an imaging plate patterned with HMA, was validated using CSC-enriched prostate and colon cancer cell lines. The HMA methodology and quantitative image analysis at single-element resolution clearly demonstrates several levels of tumor cell heterogeneity, including morphological and phenotypic variability, differences in proliferation capacity and in drug response. Moreover, the system facilitates real-time examination of single stem cell (SC) fate, as well as drug-induced alteration in expression of stemness markers. The technology may be applicable in personalized cancer treatment, including multiplex ex vivo analysis of heterogeneous patient-derived tumor specimens, precise detection and characterization of potentially dangerous cell phenotypes, and for representative evaluation of drug sensitivity of CSCs and other ty pes of tumor cells.

Donut-shaped chambers for analysis of biochemical processes at the cellular and subcellular levels
In order to study cell–cell variation with respect to enzymatic activity, individual live cell analysis should be complemented by measurement of single cell content in a biomimetic environment on a cellular scale arrangement. This is a challenging endeavor due to the small volume of a single cell, the low number of targetmolecules and cellmotility. Micro-arrayed donut-shaped chambers (DSCs) of femtoliter (fL),picoliter (pL),and nanoliter (nL) volumes have been developed and produced for the analysis of biochemical reaction at the molecular, cellular and multicellular levels, respectively. DSCs are micro-arrayed, miniature vessels, in which each chamber acts as an individual isolated reaction compartment. Individual live cells can settle in the pL and nL DSCs, share the same space and be monitored under the microscope in a noninvasive,time-resolved manner. Following cell lysis and chamber sealing, invasive kinetic measurement based oncell content is achieved for the same individual cells. The fL chambers are used for the analysis of the same enzyme reaction at the molecular level. The various DSCs were used in this proof-of-principle work to analyze the reaction of intracellular esterase in both primary and cell line immune cell populations.
These unique DSC arrays are easy to manufacture and offer an inexpensive and simple operating system for biochemical reaction measurement of numerous single cells used in various practical applications.
Monitoring gene expression in individual living cells
In this project, we transfect cells with expression vectors of the enhanced green fluorescent protein (EGFP) and with expression vectors of chimeras between EGFP and other proteins. After transfection, EGFP expressing cells are loaded onto the Cell Retainer (CR) in order to measure the fluorescent signals from each single cell and to monitor the differences between single cells in the culture. Fluorescence measurements include the kinetics of translocation and degradation of the EGFP-tagged proteins induced by a biologically relevant trigger.
A shorter procedure for the development of EGFP-expressing stable cell lines is being established by using the CR technology, by loading transfected cells onto the CR, after one week of antibiotic selection. The laser tweezers are used to unload positive EGFP-expressing cells from the CR and seed them in a fresh tissue culture dish, each cell in a single well.
Live cell sensors
This study develops a cell based methodology for simultaneous analysis of cellular functionality, secreted metabolites and molecular content at the individual cell resolution.
In this project, two sets of cells are used. One set acts as "sensing cells" for the extra-cellular medium (to sense molecules secreted or diffused from other cells), while the other set includes the tested cells that produce specific effects or molecules. The positioning of cells in the Cell Retainer can be either controlled or random. Controlled positioning of "sensing cells" is done consecutively by optical tweezers, while random positioning is a simultaneous (thus faster) procedure in which a suspension of mixed target and sensing cells is loaded onto the CR and their spatial distribution in the array is statistically determined by controlling their initial concentration ratio in the mixed cell suspension.
The resulting methodology can be applied in drug discovery, pharmacological screening, cell therapy and personal diagnosis. For example, this technique can be used in monitoring the intercellular diffusion of nitric oxide (NO) and reactive oxygen species (ROS).
Monitoring changes in membrane potential by calculating energy transfer efficiency via fluorescence polarization measurements
One of the most important functions of the cell membrane is to control material transport into and out of the cell. In most cases, control over membrane permeability is executed by changes in the membrane surface electric potential. Hence, the ability to detect these changes may provide a convenient and precise tool for monitoring membrane activity under the influence of various agents, such as drugs, mitogens, or toxins. Such monitoring tools, the correct interpretation of the measured signal, and the mapping ability in various cellular areas at an individual cell level, are of obvious importance.
In this project, a unique method is being developed to monitor the membrane potential in living cells by fluorescence resonance energy transfer (FRET) between Coumarin and Oxanol, via fluorescence polarization measurements. We measure changes in the degree of fluorescence polarization of the donor resulting from changes in membrane potential, due to which the proximity between the donor and acceptor changes. This permits its FRET mapping in various cellular areas at an individual cell level, yielding spatial information regarding membrane potential alterations.
Differential temporal and spatial biophysical aspects of the fluorescence of cellular fluorophores
In a heterogeneous environment, such as cellular media, different zones may possess various physiochemical features, which dissimilarly influence the spectroscopic characteristics of fluorescent molecules hosted in the different zones. These differently-influenced fluorophores (even from the same type) may be simultaneously monitored and eventually correlated via Time Resolved Measurements (TRM), if their fluorescence life time (FLT) - iF and/or rotational correlation time - iR differ by at least 0.5 and about 1 nsec correspondingly.
This study is the first to develop high resolution TRM-based microscopy for single cells, which will validly, quantitatively and directly probe the cellular fluorophore's local environment, e.g. refractive index, viscosity, pH, ionic strength, etc. before, during and after cell treatment.
More importantly, using both FLT and fluorescence anisotropy decay (FAD), the weight of each emitting group may be best assessed, and thus simultaneously traced, whereby the chemical-physical mechanisms involved may be interpreted and local cellular alterations can be monitored.
In the current study, TRM based microscopy is combined with the CR methodology to enable the investigation of drug - protein, and protein – protein interactions, as well as dimerisation of receptors, protein phosphorylation and conformational changes upon stimulation. For example, the mechanism of binding of the anti-inflammatory drug methotrexate with cellular proteins is investigated in lymphocytes derived from healthy donors and rheumatoid arthritis patients.
The development and utilization of Fluorescence Resonance Energy Transfer (FRET) imaging based on Fluorescence Polarization (FP) measurements
FRET has become a widely used spectroscopic tool for detecting molecular interactions and molecular proximity in solutions, as well as in membranes. Yet, FRET is generally assessed via absolute fluorescence intensity measurements, which are sensitive to the emission band used. On the other hand, FP measurement is ratiometric, simple, predictive and insensitive to inner-filter effects. Moreover, the determining of fluorescence energy transfer efficiency (E) through FP is simple to perform, conveniently adaptable to the commonly used fluorescent microscopy, and readily interpretable.
The Jerome Schottenstein Center was the first to perform FP-based FRET measurements, and we are advancing and adapting this scientific success for cell based assays, in order for that to become a routine biophysical imaging tool in High Content Analysis. As a model, FRET efficiency (E) is assessed between fluorescein and rhodamine conjugated ConA attached to receptors in the lymphocyte membrane.
This technique can be further used to monitor time and spatial dependency of essential activities in living cells, such as protein synthesis by ribosomes or post translational protein modifications, as well as protein interactions within the plasma membrane. For the study of protein synthesis and translocation, wide-field deconvolution microscopy is used; whereas total internal reflection microscopy is used to monitor processes occurring in the membrane adjacent to the Cell Retainer pico-well bottom.
Quantitative analysis of tumor metastatic potential at the individual cell resolution via diffusion characteristics
One of the most important stages in the development of primary breast cancer tissue into a secondary metastatic tissue is the dismantling of the extracellular matrix (ECM), which connects the tissue cells. In the process of breast cancer invasion and metastasis, ECM and components of the degraded basal membrane play a critical role. The matrix metalloproteinases (MMPs) are a large family of proteolytic enzymes, which are involved in the degradation of many different components of the ECM in breast cancer. MMPs occur in high levels in cancer cells and are secreted by them. The aim of this study is to quantify the "potential invasiveness†levels of cancer cells, in spreading cancer. This is done by monitoring the amount, the kinetics, the secretion, and the spread of the MMP enzyme, at a single cell resolution. First, the activity of purified MMP enzyme is measured using gelatin substrate, which is a denatured type of collagen, a major component of the ECM. We use a highly quenched gelatin substrate which, upon digestion by MMP, releases bright fluorescent peptides. The increase in fluorescence upon degradation is proportional to the proteolytic activity.
By monitoring the fluorescence time dependency, both the action rate and diffusion parameters (through the gel) can be assessed. In order to do this in a controlled environment, special geometrical arrangements of gel are molded with pre-designed symmetries, in order to simplify the analytical treatment of the differential diffusion equation under the initial and boundary conditions of these specially designed arrangements. From the comparison between the analytical solutions, derived under the specific substrate arrangement, the equation parameters are assessed and their relevancy and specificity for normal cells, cancer cells, as well as treated cells, are evaluated. Finally, similar experiments are performed directly on individual cancer cells which are positioned in the Cell Retainer in order to characterize their metastatic potential utilizing these novel individual cell-based MMP diffusion parameters. Initial results indicate the heterogeneity of cells’ MMP diffusion parameters.
Analysis of laser scattering patterns as a measure of morphological shape changes:
This study aims to conduct relatively long-term, real-time monitoring of subtle, early changes in cellular volume and conformation (due to alterations in water content, which usually precedes many other cellular events that are commonly detected by fluorophores), induced by mitogens, antigens, allergens, and hormones.
This is done by developing a single cell 'wide angle laser light scattering analyzer'. There are several advantages to such an approach. First, the use of fluorescent probes might be fully avoided (in many cases fluorophores interfere with cell physiology, bleach, quench, passively leak, etc.). Secondly, the laser light used does not interact with the cell medium it is just scattered by it, consequently enabling long term measurements. Thirdly, as we have previously shown, calculating subtle spatial dimension changes via inverse Fourier transform of the object wide angle detected power spectrum (in Fourier's plane), may be more accurate with reference to image analysis or forward scattered light of flow-cytometric analysis.
Computer-controlled Optical tweezers
In this research, a comprehensive system of computer-controlled optical tweezers is being designed and incorporated within a fluorescence inverted microscope. This hardware has the capability of supporting all experimental Cell Retainer configurations, and it is capable of trapping and relocating individual cells within the pico-wells as well as from a given pico-well to auxiliary structures, such as inlet/outlet ports or reservoirs, in combined CRs. Dedicated software controls laser intensity, beam location and coordinates over the CR, stage velocity, etc. The system is used for manipulating cells, separating individual cells from a population and inter-cellular force measurements.
Monitoring drug effects and pathological pathways in living cells by non-interfering Optical Tweezers
The trapping force of optical tweezers (OTW) depends on the Clausius-Mossotti coefficient, as well as on the elevated particle volume. The fact that low intensity light OTWs do not interfere with cell physiology, makes them a convenient long-term measurement tool for tracing alterations in cells dielectric features and volume, under bio-modulation. The principal aim of this study is to trace dielectric changes taking place upon pharmaceutical treatments or cell interactions. Beside the direct scientific value of this research, one major applicative outcome may be the addition of a non-interfering monitoring agent to the arsenal of fluorescence techniques which, unfortunately, interfere with cell physiology, especially in long term measurements.
Identifying intracellular EGFP-Bid Translocation by quantitative fluorescence measurements
Protein translocation inside the cell is a known phenomenon which characterizes many biological systems and processes (e.g. cancer). This phenomenon is currently detected by attaching fluorescent protein probes to the examined protein, and imaging via fluorescence microscopy.
This study identifies protein translocation by measuring fluorescence derivative parameters, rather than by image analysis. In this study, Jurkat T cells are transfected with enhanced green fluorescent protein EGFP-Bid. Bid is a cytoplasmic protein which translocates into the mitochondria, under stimulated apoptosis. After translocation, the EGFP-Bid concentration in the mitochondria is elevated; hence a change in mitochondrial viscosity is expected. Changes in the mitochondrial viscosity and in the EGFP-Bid concentration can cause alterations in photo-physical properties of the EGFP fluorophore, such as fluorescence polarization, fluorescence polarization decay and fluorescence lifetime. Measuring these parameters provides a simple quantitative way to identify translocation, without using cumbersome, time consuming image analysis.
The biophysical mechanism of FP alteration in EGFP expressed in living cells, following stimulation
In the current study, fluorescence polarization (FP) measurements are used to monitor the expression of intracellular proteins in living cells, under physiological and pathological conditions. In recent experiments, we found that the FP of enhanced green fluorescent protein (EGFP), expressed in Jurkat-EGFP transfected cells, was increased after treatment with the mitogen phytohemagglutinin (PHA).
The aim of this study is to understand the biophysical mechanism underlying the FP increase. Especially, the involvement of Homo-Fluorescence-Resonance-Energy-Transfer (Homo-FRET) between two EGFP molecules is examined under different conditions, such as disassembly of EGFP dimers, changes in cell volume, and changes in ion concentrations (Ca2+, Na+).
Furthermore, a protocol for quantitative measurements of FP, FP-decay and fluorescence lifetime, are established to bypass cumbersome image analysis.
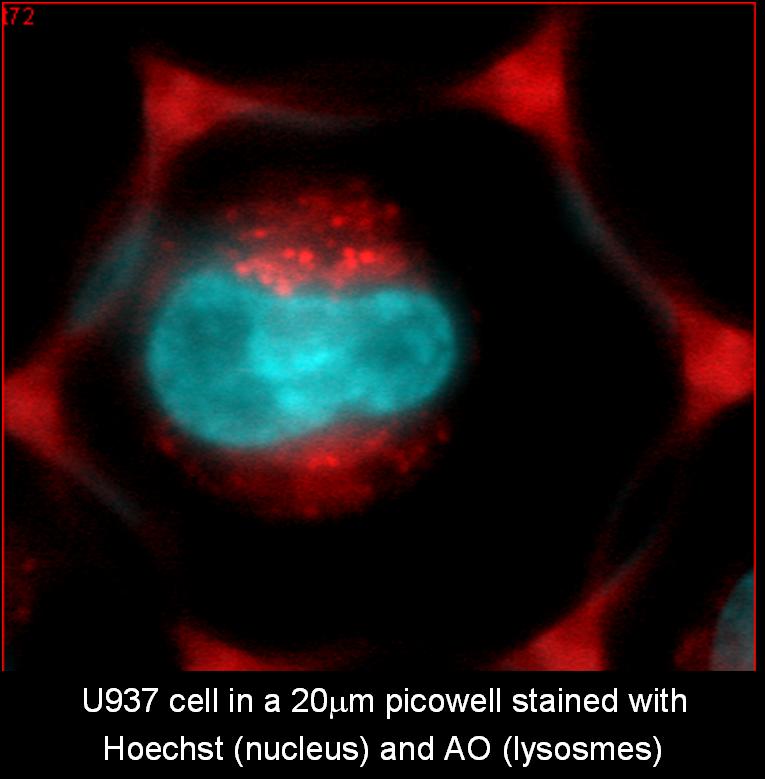
Lysosomal involvement in apoptosis induced by oxidative stress
Apoptosis induced by oxidative stress is associated with mitochondrial permeability transition and lysosomal rupture.
In this research, we use kinetic measurements of individual living promonocytic cells for a continuous monitoring of the alterations in lysosomal stability and in mitochondrial membrane potential. Lysosomal damage and mitochondrial membrane potential are detected in single cells, by observing changes in Fluorescence intensity (FI) and polarization (FP) of the vital fluorophores Acridine Orange (AO) and TMRM, respectively
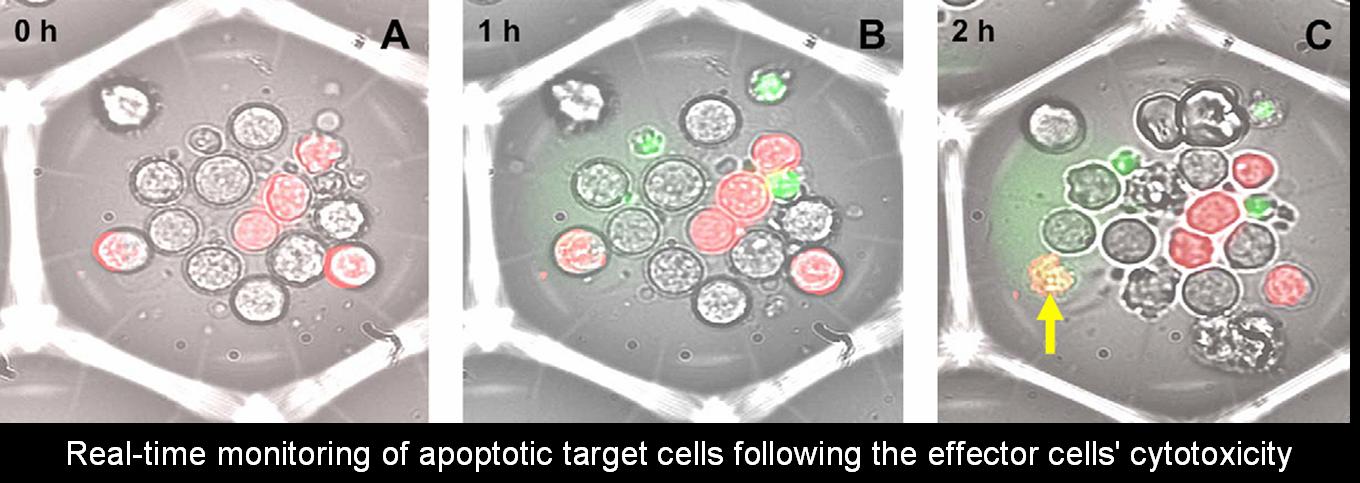
Cell-mediated cytotoxicity
Cellular cytotoxicity plays a prominent role in various physiological and pathological states. The assessment of cytolytic activity is important for monitoring immuno-competence in cancer, infectious diseases, and autoimmune diseases. Cell-mediated cytotoxicity tests are often used in mixed lymphocyte cultures, when identifying possible transplant matches.
In the current study, cytotoxicity is tested as a fundamental biochemical pathway leading to cell death/apoptosis. Utilizing the Cell Retaining methodology, real time kinetics of cytotoxicity are measured at an individual cell level. In the current model, target cells (Jurkat T cells) are co-incubated with cytotoxic effector cells (NK-92) in 100 micron Cell Retainers, and cell death is observed. In addition, immuno-phenotypic or other post-fixation analyses are conducted, to test the composition and fate of target and effector cells.